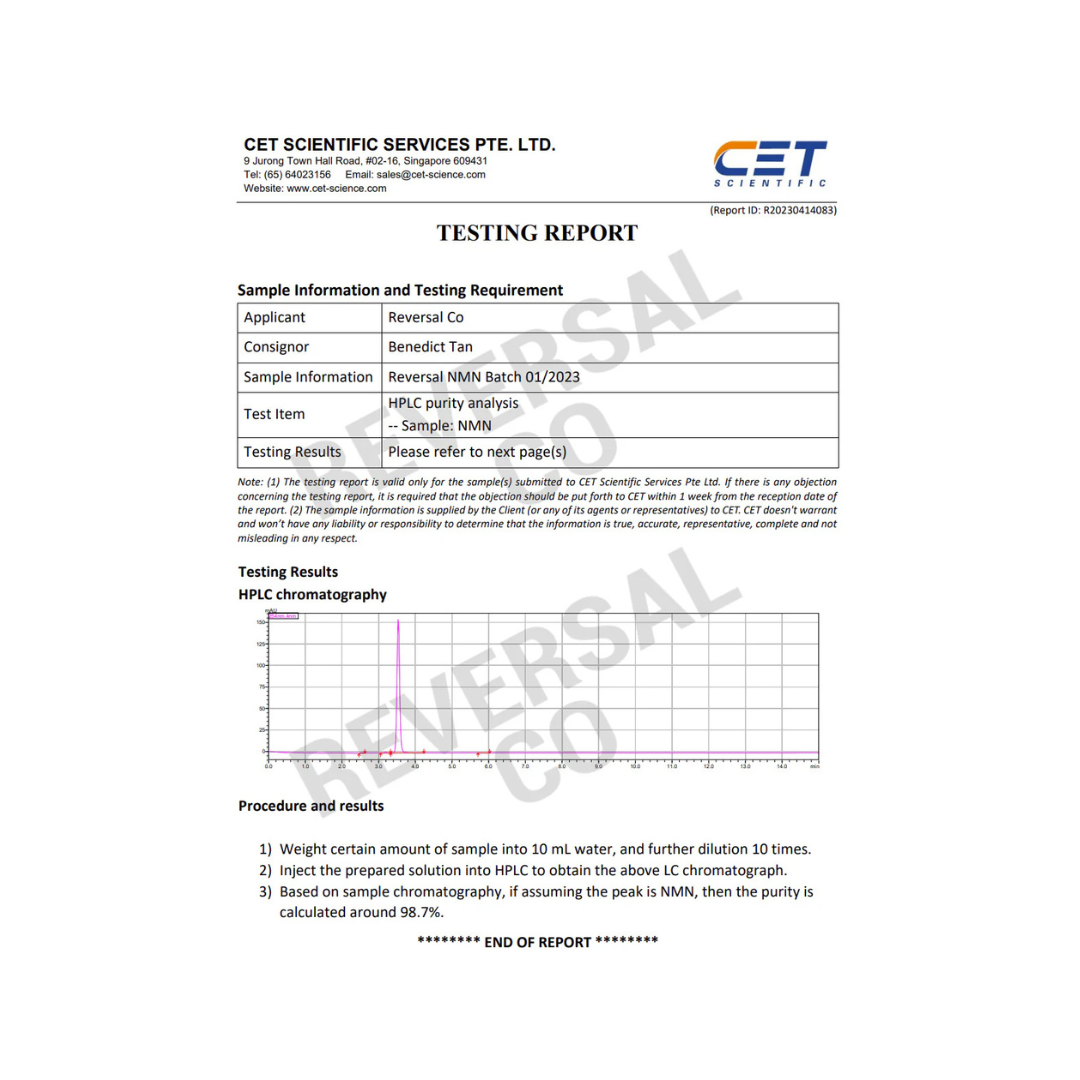
NMN Supplementation Improves Health and Increases NAD Levels in Middle-Aged Adults: Study Highlights
Introduction: A recent study has investigated the effects of β-Nicotinamide mononucleotide (NMN) supplementation on healthspan and lifespan in humans. The study examined the impact of NMN on blood NAD concentration in 80 middle-aged healthy adults. NMN is a natural compound involved in boosting NAD levels, an essential molecule for energy metabolization in mammals. Animal studies have shown promising results, thus prompting further investigation in humans.
Study Design and Objectives:
The trial utilized a multicenter, randomized, parallel, double-blinded, placebo-controlled design. The primary objective was to evaluate blood NAD concentration with varying doses of NMN. Secondary objectives included assessing the safety and tolerability of NMN, physical performance, blood biological age, insulin resistance, and overall health assessment.
Results:
Blood NAD levels increased significantly in all NMN-treated groups compared to placebo and baseline, with the 600 mg and 900 mg NMN groups showing the highest concentrations. NMN supplementation was found to be safe and well-tolerated, with no adverse effects observed. Physical performance, as measured by the six-minute walking test, improved significantly in the NMN-treated groups compared to placebo. The blood biological age increased in the placebo group but remained unchanged in the NMN groups, resulting in a significant difference between the treated groups and placebo. There were no significant differences in insulin resistance between the NMN-treated groups and placebo. The overall health assessment using the SF-36 questionnaire indicated better health in the NMN-treated groups compared to placebo.
Conclusion:
The study demonstrates that NMN supplementation up to 900 mg daily increases blood NAD concentrations, is safe, and well-tolerated. The highest clinical efficacy, indicated by blood NAD concentration and physical performance, was observed at a dose of 600 mg daily. However, further research is needed to determine the long-term effects and evaluate the impact on specific health conditions. Future studies should also explore gender differences in response to NMN supplementation.
Limitations and Future Studies:
While this study provides promising results, more comprehensive research is required. Larger studies with longer durations and additional assessments are needed to confirm the findings. Additionally, investigations into specific health conditions and potential gender differences in response to NMN supplementation are warranted.
Supplementary Information:
The trial was registered with ClinicalTrials.gov and Clinical Trial Registry - India. The supplementary material, consisting of a PDF document, provides additional information or data related to the main content of the study. It can be downloaded and viewed to gain a more in-depth understanding of the trial.
Title of paper: The efficacy and safety of β-nicotinamide mononucleotide (NMN) supplementation in healthy middle-aged adults: a randomized, multicenter, double-blind, placebo-controlled, parallel-group, dose-dependent clinical trial
Author(s): Yi L, Maier AB, Tao R, Lin Z, Vaidya A, Pendse S, Thasma S, Andhalkar N, Avhad G, Kumbhar V.
Year published: 2023
Published in: Geroscience
Original article can be found here.